Introduction
CDGs are a group of rare genetic metabolic disorders that arise from defects in the enzymatic process of glycosylation—the attachment of sugars to proteins and lipids.[1] This malfunction leads to the production of abnormal glycoproteins, which are crucial for various cellular functions.
However, glycosylation is one of the most important metabolic as well as post-translation process. Various body systems could be negatively affected. Patients often present with a wide range of clinical manifestations that may include intellectual disability, seizures, splenomegaly(enlarged spleen), recurrent infections, gastrointestinal dysfunctions, and heart-related complications like cardiomyopathy and pericarditis. [2]
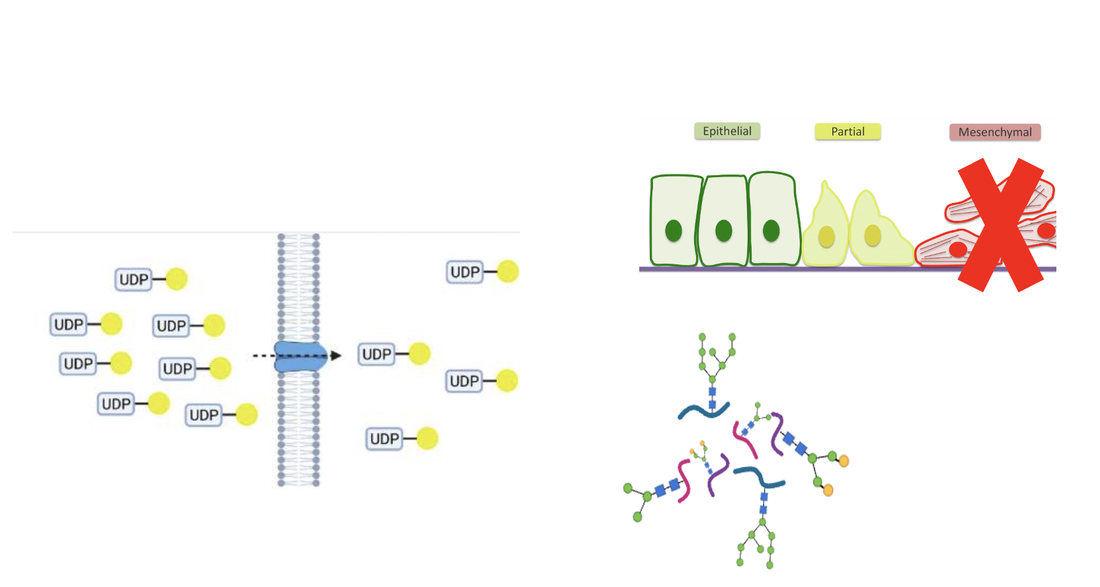
Congenital disorders of glycosylation (CDG) encompass over 170 identified genetic mutations and one specific subtype of CDG involves the inactivation of SLC35A2 genes.[1] This gene encodes for a nucleotide sugar transporter located in the Golgi apparatus. This active transporter and ion channel is important for UDP-galactose translocation from cytosol into the Golgi, which is a critical step in the glycosylation pathway. [3]
Normally, the UDP-galactose can enter the Golgi based on the concentration difference between cytosol and Golgi, proving galactose for subsequent glycosylation steps. At this point, the galactose is used for the conversion from glucosylceramide to lactosylceramide with the help of glycosyltransferase. When SLC35A2 is inactivated, there is no entry for UDP-galactose. Therefore, no available galactose within the Golgi could be used for the glycosylation pathway after the production of glucosylceramide.[4]
Kot et al. pointed out that the inactivation of SLC35A2 in epithelial cells can induce the epithelial to mesenchymal transition. The epithelial layers within GI tracts works for protecting human body from invasion of microorganisms and loss of proteins. Dysfunctions or losing of epithelial cells can causes many diseases.[5]
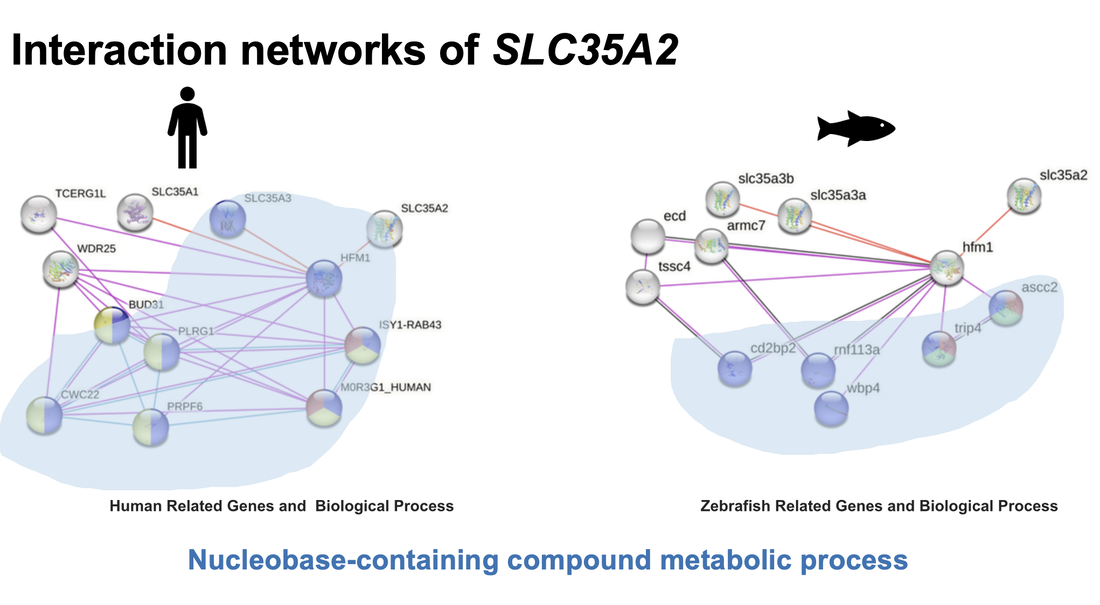
SLC35A2 is a conserved sugar transporter protein. Sequence alignments reveal that the SLC35A2 maintains highly conserved nucleotide sugar transporter domain across various species from humans plants. There are a paralogous genes found within C.elegens, named with nstp4 and srf3. Excepting nucleotide sugar transporter domain, there is an extra EamA domain within nstp4 and classed as metabolite transporters. Besides, for Arabidopsis thaliana, there is an extra UAA domain which is specific for UDP-N-Acetylglucosamine transportation. However, this pattern matches with phylogenic tree. Since C.elegans and Arabidopsis thaliana are least related with human.
There is no experimental data could be used for the SLC35A2 protein interaction analysis. But all the highlighted proteins are all related with nucleobase-containing compound metabolic process.
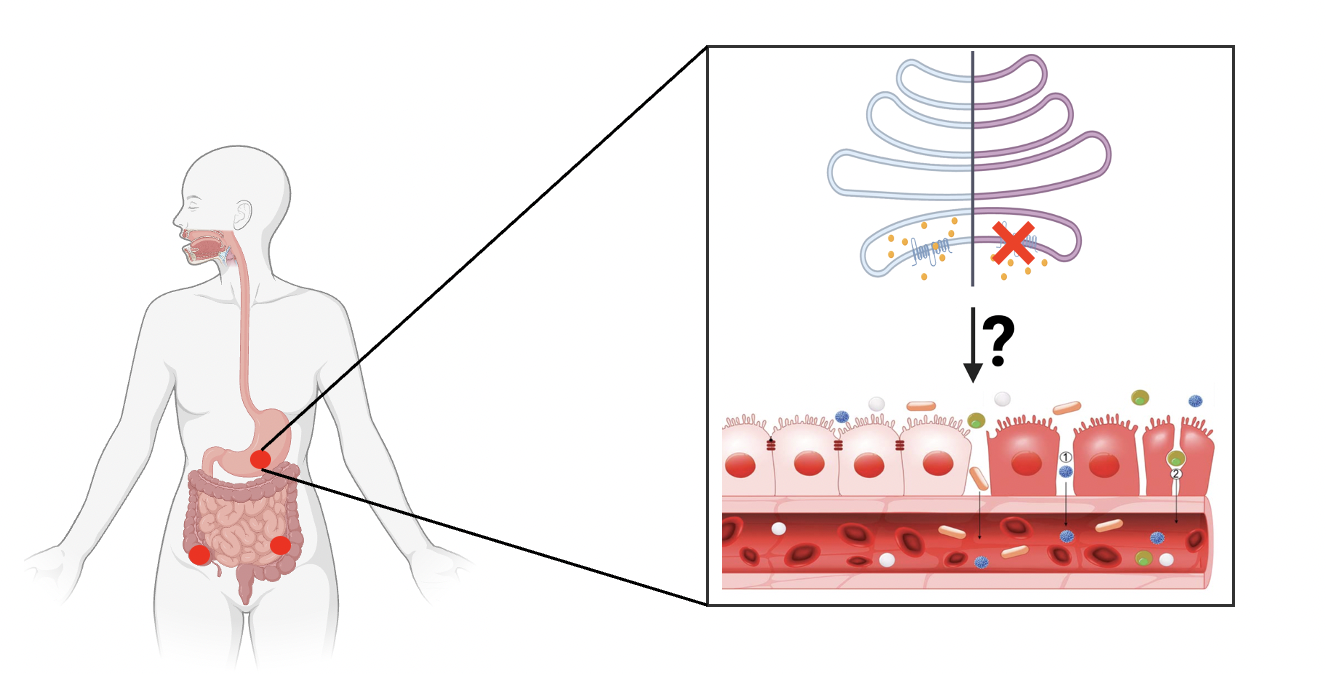
Given the genetic and physiological similarities between humans and zebrafish, coupled with the transparency features, zebrafish is an excellent model for GI tract dysfunction study. The immunofluorescence assays will be applied to visualize and quantify the extent of GI tract disorders in zebrafish. And the severity and development of the disorders could also be observed in time manner. Techniques can be readily used in the zebrafish to induce targeted mutations, knock down gene expression or create transgenic lines.
From what is hypothesized below with the inactivation of SLC35A2, the phenotype of epithelial cells within GI tract are changed. It converts from epithelial cells to mesenchymal, with loss of tight junction. Without the protective layers formed by the epithelial cells, the inflammation happened. At this time, the immunofluorescence is used to show the degree of GI disorders. Therefore, the relationship between SLC35A2 - CDG and GI tract disorders could be shown.
Despite existing studies have explored the influences of SLC35A2-CDG on various body systems, the relationship between SLC35A2 inactivation and GI tract disorder remains unclear. Specifically, the role of SLC35A2 in glycosylation of GI tract is unknown.
The primary goal is to elucidate how slc35aw glycosylation disorders on the epithelial cells lead to Gi tract disorders. There are three aims included in this project. From amino acid, protein as well as glycoproteomics level to reveal the potential relationship betwwen SLC35A2 inactivation and GI tract disorders.
AIm 1: Determining which amino acid on SLC35A2 plays an important for glycosylation in the GI tract.
Discerning each amino acid's contribution to the function of SLC35A2 can provide the framework for subsequent investigations into the roles of SLC35A2 into GI tract disorders. By using the Mega Clustal software to align sequences, several conserved amino acids have been identified, which are marked with an asterisk at the top. Previous studies have shown that mutations such as S213F and G282R can induce inactivation of SLC35A2 on human cells.[8] Therefore, the next step is to use CRISPR/Cas 9 to knockout those mutation amino acids within the proteins. Measuring the intensity of fluorescence to determine whether any disorders have occurred in the GI tract. The hypothesis here is that both mutations can inactivate SLC35A2, leading to the glycosylation disorders. Subsequently, the glycosylations disorders. Then extracting the epithelial cells from both mutant and wild type zebrafish, applying cell lysis assay to get the cytosol mixture. For HPLC analysis, the pure UDP-galactose was measured first for differrent concentration. Then based on the concentration and peak height, creating a calibration curve for peak height vs UDP-galactose concentration. The retention time should be same for each UDP-galactose concentration. And this is also the reference to identify the UDP-galactose signals when testing the cytosol samples from zebrafish models. The final UDP-galactose concentration are expected to be higher within mutant zebrafish than wild type, due to impaired utilization.
Aim2: Identifying genes related with SLC35A2 glycosylation pathway and homeostasis of GI tract.
Based on the zebrafish mutant models verified on the aim 1, the RNA sequence is used to understand how changed in gene expression due to SLC35A2 dysfunction affect the homeostasis of the GI tract epithelium. Since the changes in gene expression modulated by SLC35A2 inactivation can provide deeper insight into its roles in maintaining epithelial structure and function within GI tract. The gene expression levels from wild and mutant type zebrafish will be visualized and analyzed through heatmaps and volcano plots for better comparison and interpretation. At this point, the hypothesis is the genes associated with epithelial cells are down regulated with upregulation of mesenchymal biomarkers within mutant type zebrafish. Then the upregulated genes are sorting with Gene Ontology and KEGG pathway analysis to find out the biological process, molecular functions, and cellular components they related with. Besides, based on the KEGG analysis, the pathways they played important role in would be figured out as well. Wang et al. analyzed sequence RNA in patient's colorectal cancer. Then results can be used reference for what is expected to get from the experiments. Interestingly, one of the biological process Wang et al. found is cell fate commitment.[9] Finally, the upregulation genes are knocked out individually on both wild and mutant type zebrafish. And there is no statistical difference of fluorescence intensity are expected to be found between different zebrafish models. On the other words, knocking out trageted upregulated genes in mutant zebrafish will not induce GI tract disorders.
Aim3: Quantifying glycoproteome variations between wild-type and mutant zebrafish.
The third aim is looking forward to quantify the glycoproteins variations between mutant and wild type zebrafish. By comparing glycoprotein concentration changes across the experimental groups, providing insights into the potential therapeutic pathways or glycoproteins targets to restore normal glycosylation and maintain the intestinal integrity. There are two experimental groups, one fed with normal diet while the other treated with D-galactose enriched diet. Mutant and wild type zebrafish are involved in both experimental groups. Currently, the D-galactose is the only reported treatment for SLC35A2 - CDG, which might be able to offer some potential glycoprotein target for drug discovery.[10] Observing the fluorescence intensity change to determine the GI disorders' degree. The relief of the inflammatory response is expected on the mutant type zebrafish which feed with D-galactose enriched diet.
For the glycoproteomics part, extracting the epithelial cells from all zebrafish models. Breaking down cell membranes by either physical methods like sonication or chemical approach by using lysis buffers which contain detergents. Then extracted proteins are digested enzymatically to break them down into smaller peptides. Normal digestion or glycoside hydrolases include glycosidase, glycoproteases, proteases and trypsin. The trypsin is used to cleaves proteins at the C-terminus of lysine and arginine amino acid to reduce the complexity of protein mixture. Following the digestion, hydrophilic interaction liquid chromatography can be employed to saperate glycopeptides based on their hydrophilicity from peptides mixture. Subsequently, the glycopeptides are labeled with isobaric tag, and each sample labeled with a different tag, allowing for simultaneous analysis and comparison. Then, all labeled glycopeptides are mixed together for precise determination of the mass-to-charge ratios. The detailed information about the types of sugars present and their linkage patterned could be obtained from mass spectrometry.
Based on the MS analysis, mutant zebrafish with lower lactosylceramide and higher glucosylceramide concentration. Moreover, D-galactose treatment modulates the glycoproeomic profile in mutant zebrafish are expected. This treatment is particularly increases the lactosylceramide peak intensities identified via mass spectrometry.
For the glycoproteomics part, extracting the epithelial cells from all zebrafish models. Breaking down cell membranes by either physical methods like sonication or chemical approach by using lysis buffers which contain detergents. Then extracted proteins are digested enzymatically to break them down into smaller peptides. Normal digestion or glycoside hydrolases include glycosidase, glycoproteases, proteases and trypsin. The trypsin is used to cleaves proteins at the C-terminus of lysine and arginine amino acid to reduce the complexity of protein mixture. Following the digestion, hydrophilic interaction liquid chromatography can be employed to saperate glycopeptides based on their hydrophilicity from peptides mixture. Subsequently, the glycopeptides are labeled with isobaric tag, and each sample labeled with a different tag, allowing for simultaneous analysis and comparison. Then, all labeled glycopeptides are mixed together for precise determination of the mass-to-charge ratios. The detailed information about the types of sugars present and their linkage patterned could be obtained from mass spectrometry.
Based on the MS analysis, mutant zebrafish with lower lactosylceramide and higher glucosylceramide concentration. Moreover, D-galactose treatment modulates the glycoproeomic profile in mutant zebrafish are expected. This treatment is particularly increases the lactosylceramide peak intensities identified via mass spectrometry.
Future Work
In the future, the effective dose of the D-galactose intake could be tested. Besides, based on the mesenchymal biomarkers as well as glycoprotein variations found before, the reverse chemical screening could be applied to find whether any chemical compounds would rescue the disorders of GI tract caused by SLC35A2 - CDG.
Conclusion
The mutations at S213F and G282R can inactivate the SLC35A2 within zebrafish models. The SLC35A2 inactivation leads to the epithelial to mesenchymal transitions, by which GI tract disorders are induced. However, the intake of D-galactose can help to restore the normal glycosylation within zebrafish GI tract.
Final talk:
| chen20240229.ppt | |
| File Size: | 2858 kb |
| File Type: | ppt |
| chen20240324draft2_compress.pptx | |
| File Size: | 7039 kb |
| File Type: | pptx |
| chen20240430finaltlak.pdf | |
| File Size: | 1202 kb |
| File Type: | |
Reference:
[1]Home | CDG Hub. (n.d.). https://www.cdghub.com/
[2]Verheijen, J., Tahata, S., Kozicz, T., Witters, P., & Morava, É. (2020). Therapeutic approaches in Congenital Disorders of Glycosylation (CDG) involving N-linked glycosylation: an update. Genetics in Medicine, 22(2), 268–279. https://doi.org/10.1038/s41436-019-0647-2
[3]Consortium, G. O. (n.d.). AmiGO 2: Search. https://amigo.geneontology.org/amigo/search/bioentity?q=SLC35A2
[4]Bonduelle, T., Hartlieb, T., Baldassari, S., Sim, N. S., Kim, S. H., Kang, H. C., Kobow, K., Coras, R., Chipaux, M., Dorfmüller, G., Adle‐Biassette, H., Aronica, E., Lee, J., Blümcke, I., & Baulac, S. (2021). Frequent SLC35A2 brain mosaicism in mild malformation of cortical development with oligodendroglial hyperplasia in epilepsy (MOGHE). Acta Neuropathologica Communications, 9(1). https://doi.org/10.1186/s40478-020-01085-3
[5]Dörre, K., Olczak, M., Wada, Y., Sosicka, P., Grüneberg, M., Reunert, J., Kurlemann, G., Fiedler, B., Biskup, S., Hörtnagel, K., Rust, S., & Marquardt, T. (2015). A new case of UDP‐galactose transporter deficiency (SLC35A2‐CDG): molecular basis, clinical phenotype, and therapeutic approach. Journal of Inherited Metabolic Disease, 38(5), 931–940. https://doi.org/10.1007/s10545-015-9828-6
[6]SLC35A2 solute carrier family 35 member A2 [Homo sapiens (human)] - Gene - NCBI. (n.d.). https://www.ncbi.nlm.nih.gov/gene/7355
[7]SMART: Sequence analysis results for P78381-4. (n.d.). https://smart.embl.de/smart/show_motifs.pl?ID=P78381-4&DO_PFAM=DO_PFAM&
[8]Li, D., & Mukhopadhyay, S. (2019). Functional analyses of the UDP-galactose transporter SLC35A2 using the binding of bacterial Shiga toxins as a novel activity assay. Glycobiology, 29(6), 490–503. https://doi.org/10.1093/glycob/cwz016
[9]Wang, Y., Chen, L., Chen, J., Bai, Z., & Cao, L. (2023). Comprehensive analysis of transcriptome data and experimental identification show that solute carrier 35 member A2 (SLC35A2) is a prognostic marker of colorectal cancer. Aging. https://doi.org/10.18632/aging.205145
[10]Abuduxikuer, K. (2021). Four new cases of SLC35A2-CDG with novel mutations and clinical features. Frontiers in Genetics, 12. https://doi.org/10.3389/fgene.2021.658786
[1]Home | CDG Hub. (n.d.). https://www.cdghub.com/
[2]Verheijen, J., Tahata, S., Kozicz, T., Witters, P., & Morava, É. (2020). Therapeutic approaches in Congenital Disorders of Glycosylation (CDG) involving N-linked glycosylation: an update. Genetics in Medicine, 22(2), 268–279. https://doi.org/10.1038/s41436-019-0647-2
[3]Consortium, G. O. (n.d.). AmiGO 2: Search. https://amigo.geneontology.org/amigo/search/bioentity?q=SLC35A2
[4]Bonduelle, T., Hartlieb, T., Baldassari, S., Sim, N. S., Kim, S. H., Kang, H. C., Kobow, K., Coras, R., Chipaux, M., Dorfmüller, G., Adle‐Biassette, H., Aronica, E., Lee, J., Blümcke, I., & Baulac, S. (2021). Frequent SLC35A2 brain mosaicism in mild malformation of cortical development with oligodendroglial hyperplasia in epilepsy (MOGHE). Acta Neuropathologica Communications, 9(1). https://doi.org/10.1186/s40478-020-01085-3
[5]Dörre, K., Olczak, M., Wada, Y., Sosicka, P., Grüneberg, M., Reunert, J., Kurlemann, G., Fiedler, B., Biskup, S., Hörtnagel, K., Rust, S., & Marquardt, T. (2015). A new case of UDP‐galactose transporter deficiency (SLC35A2‐CDG): molecular basis, clinical phenotype, and therapeutic approach. Journal of Inherited Metabolic Disease, 38(5), 931–940. https://doi.org/10.1007/s10545-015-9828-6
[6]SLC35A2 solute carrier family 35 member A2 [Homo sapiens (human)] - Gene - NCBI. (n.d.). https://www.ncbi.nlm.nih.gov/gene/7355
[7]SMART: Sequence analysis results for P78381-4. (n.d.). https://smart.embl.de/smart/show_motifs.pl?ID=P78381-4&DO_PFAM=DO_PFAM&
[8]Li, D., & Mukhopadhyay, S. (2019). Functional analyses of the UDP-galactose transporter SLC35A2 using the binding of bacterial Shiga toxins as a novel activity assay. Glycobiology, 29(6), 490–503. https://doi.org/10.1093/glycob/cwz016
[9]Wang, Y., Chen, L., Chen, J., Bai, Z., & Cao, L. (2023). Comprehensive analysis of transcriptome data and experimental identification show that solute carrier 35 member A2 (SLC35A2) is a prognostic marker of colorectal cancer. Aging. https://doi.org/10.18632/aging.205145
[10]Abuduxikuer, K. (2021). Four new cases of SLC35A2-CDG with novel mutations and clinical features. Frontiers in Genetics, 12. https://doi.org/10.3389/fgene.2021.658786
Figure Reference:
Title Background: nature video. (2013, February 5). Immunology in the gut mucosa [Video]. YouTube. https://www.youtube.com/watch?v=gnZEge78_78
In-text Image:
[1] BioRender
[2]Home | CDG Hub. (n.d.). https://www.cdghub.com/
[3] https://drraghu.com/services/conditions/pericarditis/
[4]https://www.news-medical.net/health/Enlarged-Spleen-%28Splenomegaly%29.aspx
[5]Bonduelle, T., Hartlieb, T., Baldassari, S., Sim, N. S., Kim, S. H., Kang, H. C., Kobow, K., Coras, R., Chipaux, M., Dorfmüller, G., Adle‐Biassette, H., Aronica, E., Lee, J., Blümcke, I., & Baulac, S. (2021). Frequent SLC35A2 brain mosaicism in mild malformation of cortical development with oligodendroglial hyperplasia in epilepsy (MOGHE). Acta Neuropathologica Communications, 9(1). https://doi.org/10.1186/s40478-020-01085-3
[6]Nishihara, S., & Nakayama, K. (n.d.). Figure 1: [Schematic diagram of nucleotide-sugar transporter. . .]. - Glycoscience Protocols (GlycoPODv2) - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK593950/figure/g111-sugartransport.F1/?report=objectonly
[7]Premanandan, R. J. a. C. (2017, August 22). Golgi Apparatus. Pressbooks. https://ohiostate.pressbooks.pub/vethisto/chapter/1-golgi-apparatus/
[8]Nguyen, T. K., Petrikas, M., Chambers, B. E., & Wingert, R. A. (2023). Principles of Zebrafish Nephron segment Development. Journal of Developmental Biology, 11(1), 14. https://doi.org/10.3390/jdb11010014
[9]Wang, Y., Chen, L., Chen, J., Bai, Z., & Cao, L. (2023). Comprehensive analysis of transcriptome data and experimental identification show that solute carrier 35 member A2 (SLC35A2) is a prognostic marker of colorectal cancer. Aging. https://doi.org/10.18632/aging.205145
[10]Pernemalm, M. (n.d.). CANCER PROTEOMICS: METHOD DEVELOPMENT FOR MASS SPECTROMETRY BASED ANALYSIS OF CLINICAL MATERIALS. ResearchGate. https://www.researchgate.net/publication/242422798_CANCER_PROTEOMICS_METHOD_DEVELOPMENT_FOR_MASS_SPECTROMETRY_BASED_ANALYSIS_OF_CLINICAL_MATERIALS
[11]Park, J. H., & Marquardt, T. (n.d.). Treatment Options in Congenital Disorders of Glycosylation. Frontiers in Genetics, 12. https://doi.org/10.3389/fgene.2021.735348
[12]Marconi, G., Fonticoli, L., Rajan, T. S., Pierdomenico, S., Trubiani, O., Pizzicannella, J., & Diomede, F. (2021). Epithelial-Mesenchymal transition (EMT): the Type-2 EMT in wound healing, tissue regeneration and organ fibrosis. https://www.semanticscholar.org/paper/Epithelial-Mesenchymal-Transition-(EMT)%3A-The-Type-2-Marconi-Fonticoli/fb1820d20073b8e34a034fb102e4c16c491ab887
Title Background: nature video. (2013, February 5). Immunology in the gut mucosa [Video]. YouTube. https://www.youtube.com/watch?v=gnZEge78_78
In-text Image:
[1] BioRender
[2]Home | CDG Hub. (n.d.). https://www.cdghub.com/
[3] https://drraghu.com/services/conditions/pericarditis/
[4]https://www.news-medical.net/health/Enlarged-Spleen-%28Splenomegaly%29.aspx
[5]Bonduelle, T., Hartlieb, T., Baldassari, S., Sim, N. S., Kim, S. H., Kang, H. C., Kobow, K., Coras, R., Chipaux, M., Dorfmüller, G., Adle‐Biassette, H., Aronica, E., Lee, J., Blümcke, I., & Baulac, S. (2021). Frequent SLC35A2 brain mosaicism in mild malformation of cortical development with oligodendroglial hyperplasia in epilepsy (MOGHE). Acta Neuropathologica Communications, 9(1). https://doi.org/10.1186/s40478-020-01085-3
[6]Nishihara, S., & Nakayama, K. (n.d.). Figure 1: [Schematic diagram of nucleotide-sugar transporter. . .]. - Glycoscience Protocols (GlycoPODv2) - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK593950/figure/g111-sugartransport.F1/?report=objectonly
[7]Premanandan, R. J. a. C. (2017, August 22). Golgi Apparatus. Pressbooks. https://ohiostate.pressbooks.pub/vethisto/chapter/1-golgi-apparatus/
[8]Nguyen, T. K., Petrikas, M., Chambers, B. E., & Wingert, R. A. (2023). Principles of Zebrafish Nephron segment Development. Journal of Developmental Biology, 11(1), 14. https://doi.org/10.3390/jdb11010014
[9]Wang, Y., Chen, L., Chen, J., Bai, Z., & Cao, L. (2023). Comprehensive analysis of transcriptome data and experimental identification show that solute carrier 35 member A2 (SLC35A2) is a prognostic marker of colorectal cancer. Aging. https://doi.org/10.18632/aging.205145
[10]Pernemalm, M. (n.d.). CANCER PROTEOMICS: METHOD DEVELOPMENT FOR MASS SPECTROMETRY BASED ANALYSIS OF CLINICAL MATERIALS. ResearchGate. https://www.researchgate.net/publication/242422798_CANCER_PROTEOMICS_METHOD_DEVELOPMENT_FOR_MASS_SPECTROMETRY_BASED_ANALYSIS_OF_CLINICAL_MATERIALS
[11]Park, J. H., & Marquardt, T. (n.d.). Treatment Options in Congenital Disorders of Glycosylation. Frontiers in Genetics, 12. https://doi.org/10.3389/fgene.2021.735348
[12]Marconi, G., Fonticoli, L., Rajan, T. S., Pierdomenico, S., Trubiani, O., Pizzicannella, J., & Diomede, F. (2021). Epithelial-Mesenchymal transition (EMT): the Type-2 EMT in wound healing, tissue regeneration and organ fibrosis. https://www.semanticscholar.org/paper/Epithelial-Mesenchymal-Transition-(EMT)%3A-The-Type-2-Marconi-Fonticoli/fb1820d20073b8e34a034fb102e4c16c491ab887